So2 molecular geometry shape12/30/2023  
#O=S^(+)-O^-# is the standard representation proposed in texts and in schools ot chemistry. Note that the alternative Lewis structure #O=S=O# is still bent (why?), and entirely consistent with the structural parameters I have advanced. According to the VSEPR chart, the molecule which has the AX2N2 formula their molecular shape is bent and electron geometry is tetrahedral. The longer #S-O# bonds, and the more diffuse sulfur lone pair, tend to diminish the repulsive properties of the the sulfur lone pair. The actual which is only slightly compressed from the idealized trigonal planar geometry. The SO2 molecular geometry is a diagram that illustrates the number of valence electrons and bond electron pairs in the SO2 molecule in a specific geometric manner. Determine the molecular shape and polarity for SO2. Of course, the 2 oxygen atoms are entirely equivalent, and the resonance isomerism available to #O=S^(+)-O^-# does reflect this. Determine the electron geometry, molecular geometry, and polarity of SO2. A quick explanation of the molecular geometry of SO4 2- including a description of the SO4 2- bond angles.Looking at the SO4 2- Lewis structure we can see th. Since for a Group 16 atom, there should be 6 valence electrons for neutrality, the assigned electronic charges are consistent with the Lewis representation. it is a pale blue liquid in its standard state and slowly reacts with sunlight and decomposes into. 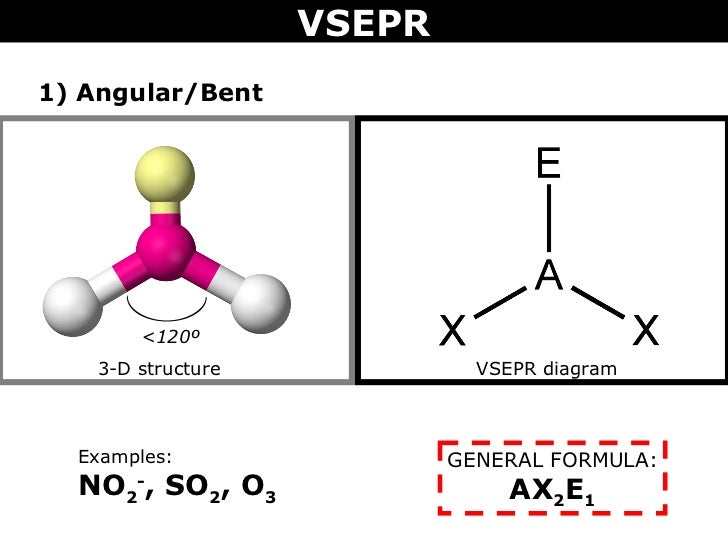
SO42- is a chemical name for the sulfate ion. There are 18 electrons to distribute in the molecule, where sulfur, as the LEAST electronegative atom, will be central.Ī Lewis structure of #O=S^(+)-O^-# is reasonable, where from left to right as we face it, there are 6, 5, and 7 valence electrons. SO2 molecular geometry is considered to V-shaped or bent. The dot structure for sulfur dioxide has sulfur with a double bond to an oxygen on the left, and two lone pairs of electrons on that oxygen, and the sulfur with a double bond to an oxygen on the right, and two lone pairs of electrons on that oxygen. SO42- Lewis Structure, Hybridization, Bond Angle and Molecular Geometry. #"VSEPR"# predicts that sulfur dioxide should be a bent molecule, where, to a first approximation, Why? 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
